February 2008 // Volume 46 // Number 1 // Feature Articles // 1FEA5
Assessment of Producer Implementation of Pork Quality Assurance Good Production Practices
Abstract
The study reported here was undertaken to measure the implementation rate of best management practices recommended by the Pork Quality Assurance (PQA) program. Data were collected via face-to-face interviews and observations with 78 pork producers in four states. The level of compliance regarding physical activities required by the PQA program was about 88%. But only about 48% of producers maintain adequate continuing education and documentation of production practices. We conclude that a sizable majority of producers perform the physical tasks needed to produce wholesome pork, but there are opportunities for improvement in regard to documentation of medication administration.
Introduction
In 1989, the National Pork Producers Council (NPPC) introduced a voluntary program for pork producers across America aimed at producing safe, wholesome pork products for consumers in both domestic and export markets. The program, Pork Quality Assurance (PQA), outlined 10 Good Production Practices (GPPs) for pork producers to implement on their farms. The 10 practices include:
- The identification and recording of all treated animals,
- Proper drug administration techniques
- Understanding drug labeling
- Adopting a valid veterinarian/client/patient relationship,
- Developing an effective herd health management plan
- Processing on-farm feed appropriately, and
- Utilizing proper swine care and handling.
After receiving initial Level III certification in the PQA program, producers were required to re-certify every 2 years. Methods to conduct PQA producer education have included a review of the 10 GPPs by an educator (veterinarian, Extension agent, or agricultural educator) in group meetings or via on-farm consultation utilizing video, slide presentation, or textbook instruction. Re-certification instruction has included a review of important GPP guidelines, videos, and other visual aids.
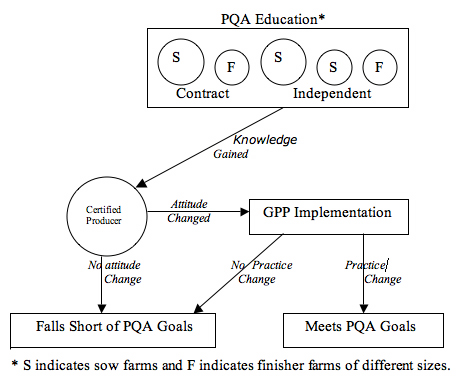
The Pork Quality Assurance program provides an educational framework for teaching producers the essential elements of producing wholesome and safe pork. As presented graphically (Creswell, 1994) in Figure 1, the goal of the program is to increase knowledge of the producer. The education model shown in Figure 1 assumes that pork producers, regardless of size or financial structure, have an opportunity to gain knowledge from the PQA training and adopt practices outlined in the program. If education and certification promote attitude change, GPPs are more likely implemented.
Figure 1.
PQA Educational Influence on
Attitude, Practice Change, and GPP
Increased pressure from consumers and food service associations upon suppliers of meat products may lead to examining the need for a voluntary assessment program. To that end, the National Pork Board is expected to develop a voluntary assessment program that would involve on-farm visits by trained professionals to evaluate the extent to which producers have implemented all PQA Good Production Standards and to be paid for by market support.
Procedures
The purpose of the study reported here was to measure the degree of implementation of the 10 Good Production Practices (GPPs) in the PQA program, which include:
- Identify and track all treated animals
- Maintain medication and treatment records
- Properly store, label, and account for all drug products and medicated feeds
- Use a valid veterinarian/client/patient relationship
- Use proper administration techniques
- Use drug residue tests when appropriate
- Establish and implement an efficient/effective herd health management plan
- Provide proper swine care
- Follow appropriate feed manufacturing procedures
- Maintain PQA certification
Swine producers with operations in the United States of America comprise the population for the study. Criteria-based sampling (Issac & Michael, 1997) was employed, with the unit of analysis being an individual producer. The target sample size was 80 producers representing five different production types, which were selected to correspond to the typical production and financial arrangements used on swine enterprises in the United States. An inventory of 300 sows was chosen as a threshold value to divide small and large independent sow operations. The five production types are presented below:
- Small independent sow operator (< 300 sows)
- Large independent sow operator (> 300 sows)
- Independent grow-finish, wean-to-finish, or nursery operator
- Contract grow-finish, wean-to-finish, or nursery operator
- Contract sow operator
To address the research objectives, a survey instrument was developed using a combination of yes/no questions, open-ended questions, categorical data collection, and the observation of quality assurance procedures by trained technicians. Our primary goal in designing the survey was to determine the extent to which swine producers had implemented each of the GPP's in the day-to-day operations of their swine enterprises. In most cases, answers to questions were recorded as (A) acceptable, (N) needs improvement, or (NA) not applicable. To record an answer as acceptable, the respondent would have to meet all criteria described in the PQA manual for each respective item.
Data were collected for this survey from 78 pork producers on farms in Minnesota (n=20), North Carolina (n=20), Oklahoma (n=18), and Pennsylvania (n=20). Trained technicians contacted participants by telephone using a uniform phone script that explained the purpose of the survey, requested consent for participation, and explained the conditions of privacy. Technicians conducted face-to-face interviews over a 5-month period with the 78 swine producers who collectively fulfilled the five categories of swine operations previously described.
Results
The results provided herein highlight the relevant components of each production practice in order to assess the overall implementation rates associated with the 10 Good Production Practices in the PQA program (Table 1).
| Good Production Practice | Adoption Rate, % |
| Identify and track all treated animals | |
| Animal identification complies with medication withdrawal | 86 |
| Maintain medication and treatment records | |
| Records meet all requirements of FDA CPG 7125.37* | 55 |
| Properly store, label, and account for all drug products and medicated feeds | |
| Storage conditions are appropriate for: | |
| Medications not requiring refrigeration | 99 |
| Medications requiring refrigeration | 73 |
| Medications requiring below-freezing temperatures | 99 |
| Outdated medications (disposed of appropriately) | 80 |
| Use a valid veterinarian/client/patient relationship (VCPR) | |
| Producers with an acceptable rating | 81 |
| Use proper administration techniques | |
| Injections administered in proper location | 96 |
| Appropriate needles used for injections | 91 |
| Water medications mixed according to label | 100 |
| Use drug residue tests when appropriate | |
| Veterinarian is consulted when drug residue tests are used | 50 |
| Establish and implement a herd health management plan | |
| Effective biosecurity practices | 68 |
| Effective rodent control program | 83 |
| Disease prevention plan is documented in writing | 53 |
| Provide proper swine care | |
| Proper restraint used during administration of medication | 82 |
| Equipment used for moving hogs minimizes stress | 92 |
| Procedure for disposal of sharps is appropriate and safe | 99 |
| Follow appropriate feed manufacturing procedures | |
| Written records of medicated feed production and purchases | 72 |
| Feed medications used according to label directions | 97 |
| Bins and feeders properly identified | 91 |
| Maintain continuing education | |
| PQA certification and continuing education up to date | 79 |
| * Requires producers to include: date administered, animal identification, product name, amount of drug given, who administered the medication, and the drug withdrawal days. (The most common reason for not completely meeting the requirements was a failure to list who administered the medication.) | |
Identification
Eighty-six percent of the producers utilized an identification system that enabled proper medication withdrawal.
Medication Records
Fifty-five percent of producers maintained written medication records that met the Food and Drug Administration (FDA) Compliance Policy Guide (CPG) 7125.37 in all respects. The FDA CPG 7125.37 requires treatment records to include: date administered, animal identification, product name, amount of drug given, who administered the medication, and the drug withdrawal days. While most producers included the required information, interviewers discovered that many producers did not include the name of the individual administering the medication.
Medication Storage
Ninety-nine percent of the producers stored non-refrigerated medications according to label directions. Seventy-three percent properly stored medications requiring refrigeration, and 99% properly stored medications requiring below freezing temperatures. Outdated medications were disposed of properly by 80% of the producers.
Veterinarian/Client/Patient/Relationship
The PQA Program requires a valid Veterinarian/Client/Patient Relationship (VCPR), which implies that the veterinarian: 1) has assumed the responsibility for making medical judgments regarding the health of the animals; 2) has sufficient knowledge of the animals; and 3) is readily available for follow-up consultations. Overall, 81% of participants in this study earned an acceptable rating for meeting the terms of a valid VCPR. Of those not meeting all the VCPR criteria, many relied on the company personnel servicing the unit (which may have a VCPR in place that was not identified due to the limitations of the survey) to help them make medical decisions.
Administration Techniques
Ninety-six percent of the producers administered injections in the proper location, and 91% utilized appropriate needles. One hundred percent of the subjects reported mixing water medications according to label.
Drug Residue Tests
Drug residue tests may be appropriate on swine farms when cull sows or young pigs are sold, or when animals are used for exhibitions. It is advisable to consult the herd veterinarian when these tests are used even though the tests are available over the counter. In this group of producers, 62 of the 78 producers had used drug residue tests; 50% of these individuals consulted their veterinarian before doing so.
Herd Health Plan
Effective herd health plans require biosecurity and rodent control procedures to minimize the spread of disease within and from other swine operations, as well as an established, written disease prevention plan. About 68% of producers in this group had effective biosecurity plans, 83% had effective rodent control programs, while 53% had documented their disease prevention plans in writing.
Proper Swine Care
Proper swine care involves a wide range of husbandry skills and appropriate equipment and facilities. Proper animal restraint was achieved by 82% of the participants, 92% used equipment for moving hogs that minimized stress, and 99% of the participants successfully prevented physical hazards related to needles and sharp objects.
Feed Handling
A sizable majority of producers handled feed in a safe and appropriate manner, as 97% used feed medications according to label directions, and 91% properly identified bins and feeders. Seventy-two percent of subjects maintained written records of medicated feed production and purchases
Continuing Education
Producers are expected to maintain their PQA certification requirements. Seventy-nine percent of producers received acceptable scores for continuing education and PQA recertification.
Conclusions
From this assessment of five different types of pork production operations that are representative of the United States swine industry, we found that pork producers, in general, successfully complete physical activities associated with the 10 Good Production Practices of the Pork Quality Assurance program. Identification systems for animals, storage conditions of medications, maintaining a good working relationship with the herd veterinarian, techniques for administering medications, animal care, and on-farm feed manufacturing procedures received high marks for this group of pork producers. This is an important finding, as these factors comprise the essential activities needed in the production of safe and wholesome pork.
There are opportunities for PQA educators to help producers improve in areas of record-keeping, documentation of production practices, and continuing education. For example, 45% of producers did not include all of the items outlined in the FDA CPG 7125.37 in their medication records (date administered, animal identification, product name, amount of drug given, who administered the medication, and the drug withdrawal days). Who administered the drug was a common omission from medication records. Pork producers establish organized disease prevention plans as a standard operating practice, but 47% of individuals in this survey group needed to document their plan in writing. Finally, producers have a wide range of continuing education opportunities, including PQA recertification, and a majority (79%) received acceptable ratings, but PQA educators could devote more attention to this area.
Educational Implications
For Extension educators serving in the role as PQA educators for the National Pork Board's Pork Quality Assurance program, the following recommendations are suggested to improve program delivery methods and the compliance levels of certified pork producers. Recommendations are listed in the order of the Good Production Practices (GPPs).
Identification
The identification of animals on sow farms is at a high level of compliance for GPP#1. However, the swine industry would benefit from more lasting methods that would serve to identify nursery, weaner, grower, and finisher pigs for medication withdrawal to slaughter.
Medication records
The PQA program provides sample record-keeping and management forms to aide producers in meeting the requirements of this GPP. It is recommended that PQA educators emphasize keeping written medication records that meet all of the items outlined by FDA. It would be helpful to have revised sample record sheets, highlighting the required items. Educators and/or NPB could produce and distribute record notebooks that satisfy the FDA requirements and prompt producers to keep appropriate records.
Medication Storage
Swine producers, in general, meet the requirements for medication storage. An opportunity for improvement is that of refrigeration temperatures. Producers do store medications in a refrigerator according to label requirements, but those who did not receive an acceptable score in this category typically had a refrigerator that was not set at the proper temperature. This provides an opportunity for educators to emphasize that refrigeration temperatures affect the shelf life of drugs and medications.
Veterinarian/Client/Patient/Relationship
Eighty-one percent of the participants in the study were in compliance with the VCPR requirements. Those who do not have a VCPR often rely on service personnel from the contracting company (which may have this relationship which not identified due to the limitations of this survey) or themselves for making medication decisions. Educators should clarify that producers need to consult with their veterinarian any time animal health products, even over-the-counter drugs, are used. The herd health plan, developed in consultation with the veterinarian, will outline a wide variety of circumstances requiring special treatment of animals. This provides a valuable day-to-day operating procedure that enables the producer to make timely treatments that are in accordance with professional veterinary judgment.
Administration Techniques
Producers appear to be administering medications appropriately as evidenced by the relatively high scores earned in this category. Educators need to remind producers that all workers and family members should be trained in using proper administration techniques as new personnel take on this responsibility.
Drug Residue Tests
About 79% of producers in this survey group used drug residue tests at one time or another. This was somewhat unexpected, but reassuring that a relatively high percentage of producers would take advantage of this tool to verify the absence of drug residues. It's important to note that educators should encourage producers to consult their veterinarian to help decide if and when such tests are needed and appropriate, and also to help investigate the causes of any positive tests that may arise.
Herd Health Plan
Pork producers typically establish an organized disease prevention plan for their farm, and this group was no exception. However, only half of the producers interviewed had their plans documented in writing. PQA educators should encourage all producers to develop written Disease Prevention plans for producers in their production systems. Written plans, by their nature, convey a stronger commitment to good herd health.
Proper Swine Care
Producers in this group exhibited good husbandry practices and achieved high scores for swine care. The NPB has developed new training materials (Swine Welfare Assurance Program - SWAP) in this important area. Producers should be encouraged by PQA educators to enroll in this program as a complement to the PQA program.
Feed Handling
Producers who process their own feed should be grouped together for PQA training because they have different GPP implementation requirements than those who purchase all feedstuffs. In the study reported here we found that producers manufacturing their own feed followed best management practices as demonstrated by the high scores associated with following label directions and proper identification of bins and feeders. Producers purchasing feed should ask for written documentation from their feed manufacturer regarding their implementation of Good Manufacturing Practices outlined in the PQA manual.
Continuing Education
Because the PQA re-certification requirement has been changed to every 3 years, it is important that producers complete their own PQA self-assessment annually. If an assessment such as the one implemented here were to become available, we estimate an average assessment time of about 2 hours (Table 2).
| Operation Type | Average Time | Range (minutes) |
| Small independent sow (n=15) | 1 hour, 39 minutes | 60 - 165 |
| Large independent sow (n=16) | 2 hours, 13 minutes | 25 - 240 |
| Independent G/F, W/F or N* (n=14) | 1 hour, 36 minutes | 26 - 150 |
| Contract G/F, W/F, or N* (n=16) | 1 hour, 52 minutes | 75 - 150 |
| Contract sow operation (n=16) | 2 hours, 12 minutes | 60 - 220 |
| All Operations (n=77) | 1 hour, 55 minutes | 25 - 220 |
| *G/F = grower/finisher, W/F = wean-to-finish, and N = nursery. | ||
Acknowledgements
The authors thank the National Pork Board for their financial support and assistance in completing this project. We are also grateful to the following individuals who assisted in the assessment of the participant farms: Bill Crawford (Minnesota), Bob Koehler (Minnesota), Kathy Dugan (North Carolina), Star Jackson (North Carolina), Bill Luce (Oklahoma), and Robert Mikesell (Pennsylvania).
References
Creswell, J. W. (1994). Research design: Qualitative & quantitative approaches. Thousand Oaks, CA: Sage.
Issac, S., & Michael, W. B. (1997). Handbook in research and evaluation (3rd ed.). San Diego,CA: EdITS.
National Pork Board. (2003). Pork quality assurance assessment educator's guide. Des Moines, IA
National Pork Board. (2002). Quality assurance: A program of America's pork producers, Level III. Des Moines, IA
